These Sensor Materials are Changing How We Monitor Health (The MedTech Download)

Key Takeaways
- Demand for, and innovation surrounding, sensor technology in healthcare has grown, with innovators related to new materials and methods for measurement.
- As the use of wearable medical technology continues to grow, neurological disorders have emerged as another category that benefits from these advancements.
- Philips recent Class 1 recall of several respiratory technologies has left several patients, even months later, without an alternative.
Sensor and Sensitivity: The Materials Changing Medical Measurements
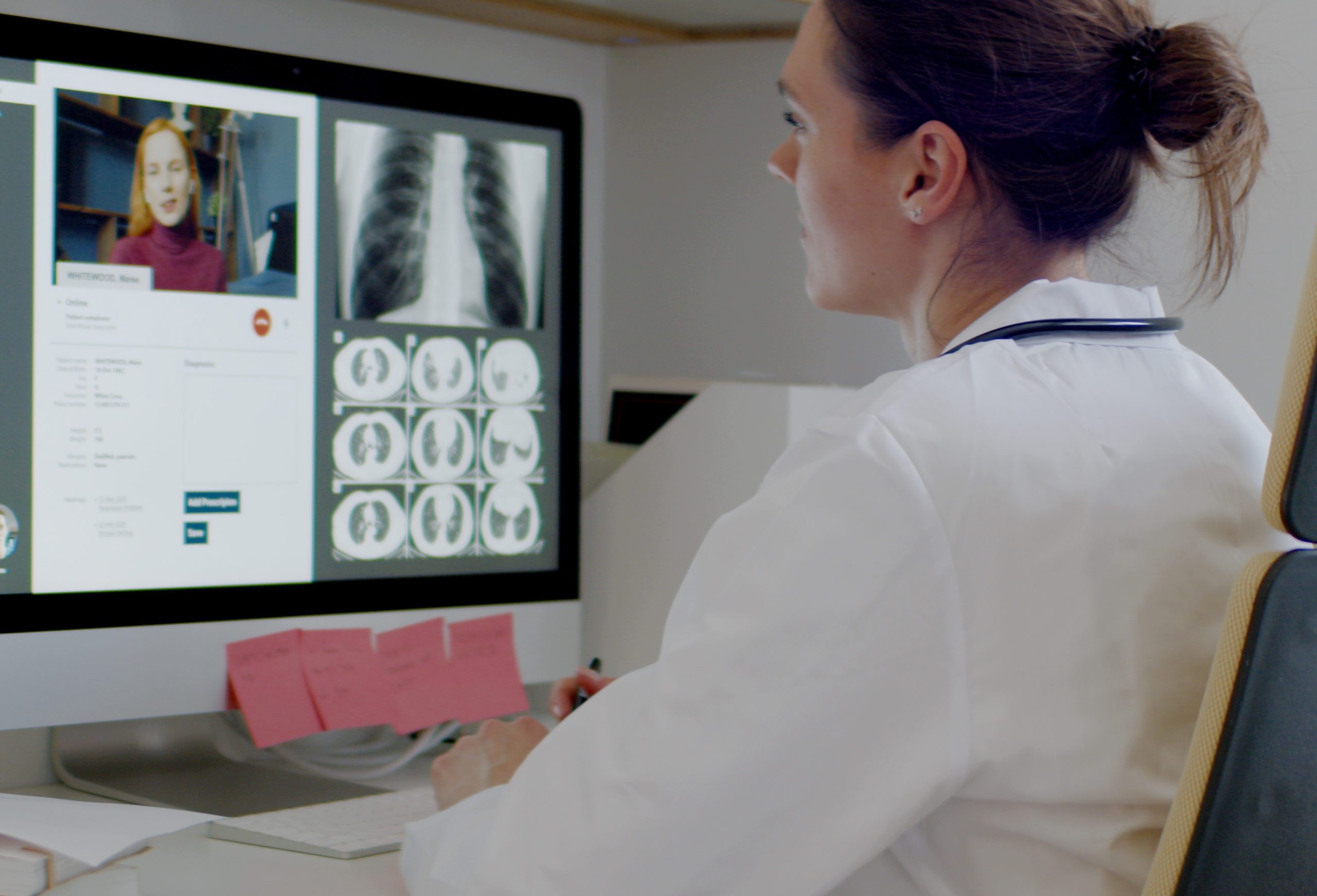
Sensors have long been an important part of healthcare delivery. From heart rate and blood glucose level to body temperature and blood pressure, monitoring vitals provides physicians with the data they need to make healthcare decisions for their patients. And now in the wake of COVID-19, reliable sensors that allow providers to monitor and treat patients remotely has ushered in a new era of demand for accurate and durable sensing technologies.
And as in any field, when there is demand, innovators will no doubt be there to provide solutions. An article published by Medical Device Network explores a few of the most recent needs and methods related to sensor technology. Some listed include a facemask sensor to detect COVID-19, railway monitoring fiber optics that have been repurposed for use in the human body, and more. You can read the full article here.
Low-Cost Nanobodies from Alpacas are Highly Potent Inhibitors of SARS-CoV-2

For the first few months of the pandemic, the medical world was not equipped to effectively treat COVID-19 patients. As we scrambled to get a grasp on identifying effective therapies, many potential treatments came and went, most memorably hydroxychloroquine, which was soon found not to be effective in treating patients infected with the virus. Now, we have vaccines, which as we all know help our bodies to produce antibodies to fight the virus, and are by far the most effective way to reduce severe disease.
Yet, there are patients who are unable to receive the vaccine due to age, health status, or choice, so healthcare providers still need tools to combat the virus in infected people. Researchers in Germany have used alpacas to engineer mini-antibodies, called nanobodies, against SARS-CoV-2. The article explains that “the team’s nanobodies are stable up to 95 degrees Celsius (203 F) and are cheaper and less complex to produce than conventional antibodies. So far, the nanobodies have shown impressive efficacy against the virus and its variants in the lab.”
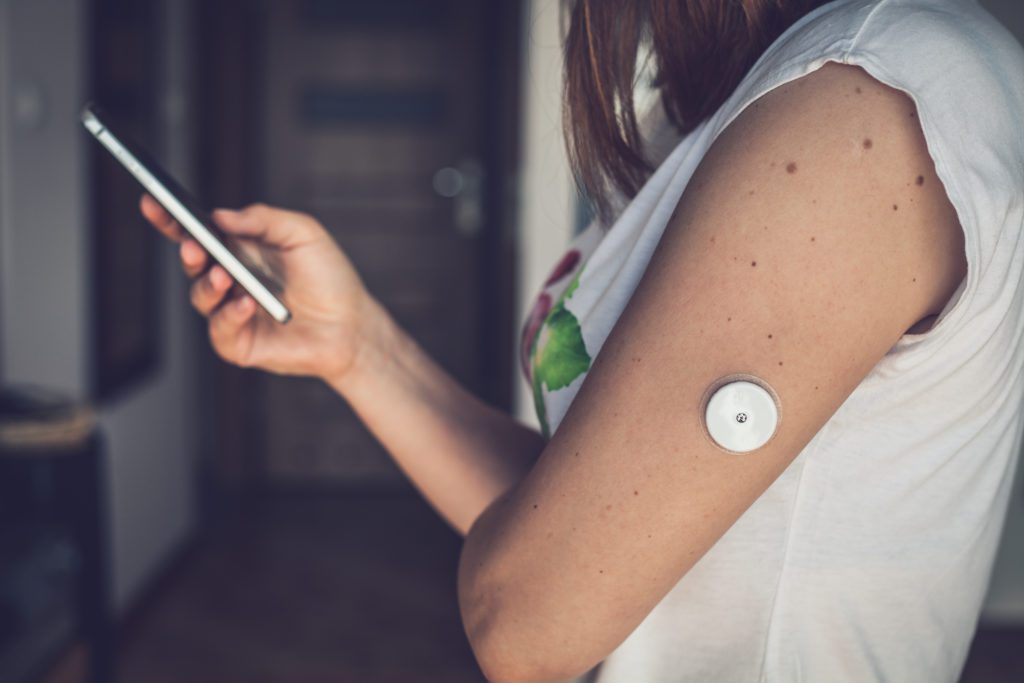
The Prevalence of Neurological Disorders Pushes Scope of Wearable Medical Devices

One of the largest growing categories in medical devices is wearable technology. Earlier this year, it was reported that, despite an earnings downturn for medical devices due to the pandemic, wearable diabetes technology seemed to be immune to the dip, with companies, including Dexcom, Abbott, and Insulet, announcing increases in sales. Since then, investment in wearable medical devices has continued to increase, as consumers with and without chronic illness continue to show interest in health monitoring.
But as most of the conversation in medical health monitoring and wearables surrounds fitness, diabetes, and cardiovascular conditions, one area in which wearables are growing is in relation to neurological disorders. As an article in MedTech Intelligence points out, “the need for equipment that monitor neurological conditions or help in the early detection or diagnosis of these diseases is greater than ever. WHO has estimated the worldwide occurrence of neurological health problems could reach nearly 103 million by 2030.”
A Recall of Philips Respiratory Devices has Left Users Stranded
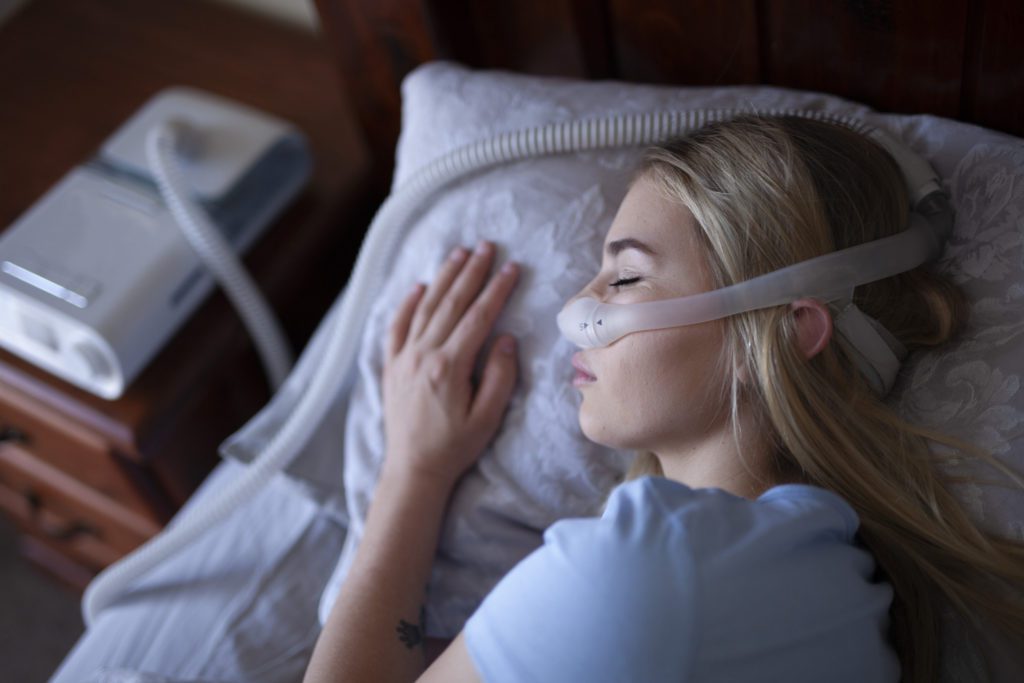
For many patients, medical technology keeps them alive. For those who rely on ventilators, CPAPs, and BiPAPs to breathe, the thought of not having access to these machines is terrifying. As Ingrid Tischer points out in an article in The Verge, “(faulty) respiratory equipment is not like a car that’s faulty,” says Tischer. “You can stop driving the car, but you can’t just postpone breathing.”
Relying completely on technology leaves people tremendously vulnerable, which is why Philips’ recent Class 1 recall of those very same respiratory devices has left millions of patients in a difficult situation. What’s worse, is that a month later, many patients still do not have a resolution. Many are forced to continue to use these devices, which carry with them a reasonable probability that use “will cause serious adverse health consequences or death.”
Stay up-to-date on everything that matters to MedTech entrepreneurs and executives, from opportunities within software as a medical device to fundraising. Subscribe below to receive industry insights, MedTech resources, and more delivered directly to your inbox.
The medtech news you want – and use – delivered. Subscribe below.