New Survey Reveals Physician Skepticism Towards Wearable Devices (The MedTech Download)

Key Takeaways
- While consumer adoption of diabetes and other healthcare wearables is on the rise, a recent survey shows that physicians are generally skeptical about using wearable technology in treating patients.
- mRNA goes well beyond vaccines – researchers in Tokyo are working on using mRNA to help stroke patients achieve better outcomes by reducing cell death.
- The Medical Device Manufacturers Association has joined 39 healthcare stakeholders in opposition to the expansion of Medicare prior authorization requirements related to cervical fusion with disc removal and implanted spinal neurostimulators.
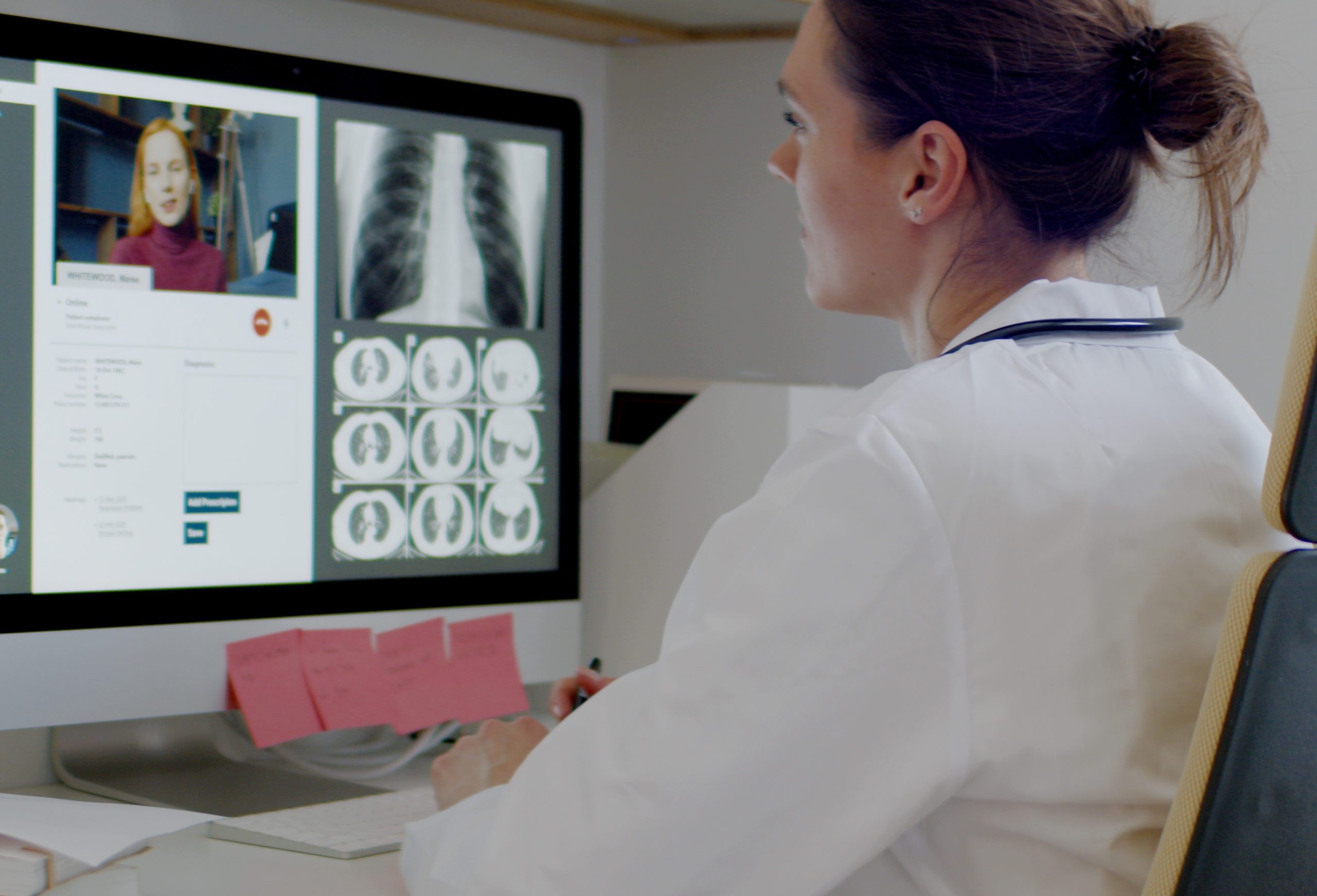
Survey Casts Doubt on Utility of Wearable Devices in Healthcare

For the diabetes community, wearable technology has been a welcome and growing trend. The disease requires near-constant monitoring of blood sugar levels and subcutaneous insulin delivery, so devices like the continuous glucose monitor (CGM) and insulin pump both simplify and optimize management. Outside of diabetes, companies are investing in the development of health monitoring wearables, from the Apple Watch to cuffless, wearable blood pressure technology. Everywhere you look, consumers are offered another wearable gadget that can help them manage their health.
However, while consumers seem optimistic about healthcare wearables, a new survey indicates that physicians feel that “consumer technology and data may never play a leading role in helping physicians provide healthcare.” Physician objections to using wearables to treat patients include mistrust in the data they provide, with one doctor stating that “companies would need to publish large studies documenting the accuracy of wearables across populations.” Read the full article here.
mRNA Delivery System Protects Neurons After Stroke

When the efficacy of both the Moderna and Pfizer vaccines were announced late last year, the results were stunning. When both reported more than 90% efficacy in preventing COVID-19, many people did a double-take. Each year, the flu vaccine provides between 40% and 60% protection against infection, so, in comparison, the results seemed close to miraculous. As we learned more about these two vaccines, we also learned about the use of synthetic messenger RNA, not only in vaccines, but beyond. And while the technology has received attention due to the current crisis, the history of synthetic mRNA is much richer.
Now researchers in Tokyo have developed a method to use mNRA “to produce brain-derived neurotrophic factor (BDNF), which can protect neurons from ischemia” following a stroke. An article in Medgadget goes on to say that “researchers hope that the treatment could help patients who have suffered a stroke to achieve better outcomes by reducing cell death.” Rat trials have shown promising results, wherein researchers noted that “the treatment worked best when it was administered two days after the ‘stroke’ rather than right away, suggesting that it might provide an extended window for effective therapy.”
Medtech Industry, Surgeons Push Medicare to Pull Back Prior Authorization Rules

The debate surrounding access to healthcare in the United States has grown over the past year. In large part, it has been spurred on by data surrounding COVID-19 mortality rates, as people of color were up to 2.4 times more likely to die from the disease than their counterparts. Included in the debate is physical access to healthcare facilities, the high costs of insurance premiums or deductibles, and issues surrounding coverage, to name a few. Included in the insurance coverage ecosystem are Medicare requirements for prior authorization for treatment.
Recently, CMS announced the addition of two new service categories in the 2021 Outpatient Prospective Payment System and Ambulatory Surgical Center final rule. The addition requires Medicare patients to receive prior authorization for cervical fusion with disc removal and implanted spinal neurostimulators. As a result, the Medical Device Manufacturers Association “joined 39 other healthcare stakeholders in voicing their strong opposition to the expansion of Medicare prior authorization requirements,” arguing it will delay patient access to procedures that are medically necessary.
NIRVANA Test Rapidly Identifies and Sequences COVID-19 Viral Variants

As COVID-19 emerged last year, our first priority was understanding how it spreads and preventing infection. Since then, the next element in getting the virus under control is figuring out how it has changed, and how the variants are impacting our communities. What is referred to as the UK variant has proven to be more contagious, yet apparently not more likely to cause severe disease. But, as the world continues to see a high-rate of infection, more variants are not only possible, they are expected. Tracking and understanding them will be key to getting back to pre-pandemic life.
Recently, researchers at the Salk Institute in California, in partnership with outside collaborators, developed a new test called the nanopore sequencing of isothermal rapid viral amplification for near real-time analysis, or NIRVANA. This novel COVID-19 test “can rapidly identify and sequence the causative virus, helping to track new variants” and can also test for other viruses that might cause similar symptoms, like the flu. The test is also small and portable, providing results within about 20 minutes.
Stay up-to-date on everything that matters to MedTech entrepreneurs and executives. Subscribe below to receive industry insights, MedTech resources, and more delivered directly to your inbox.